 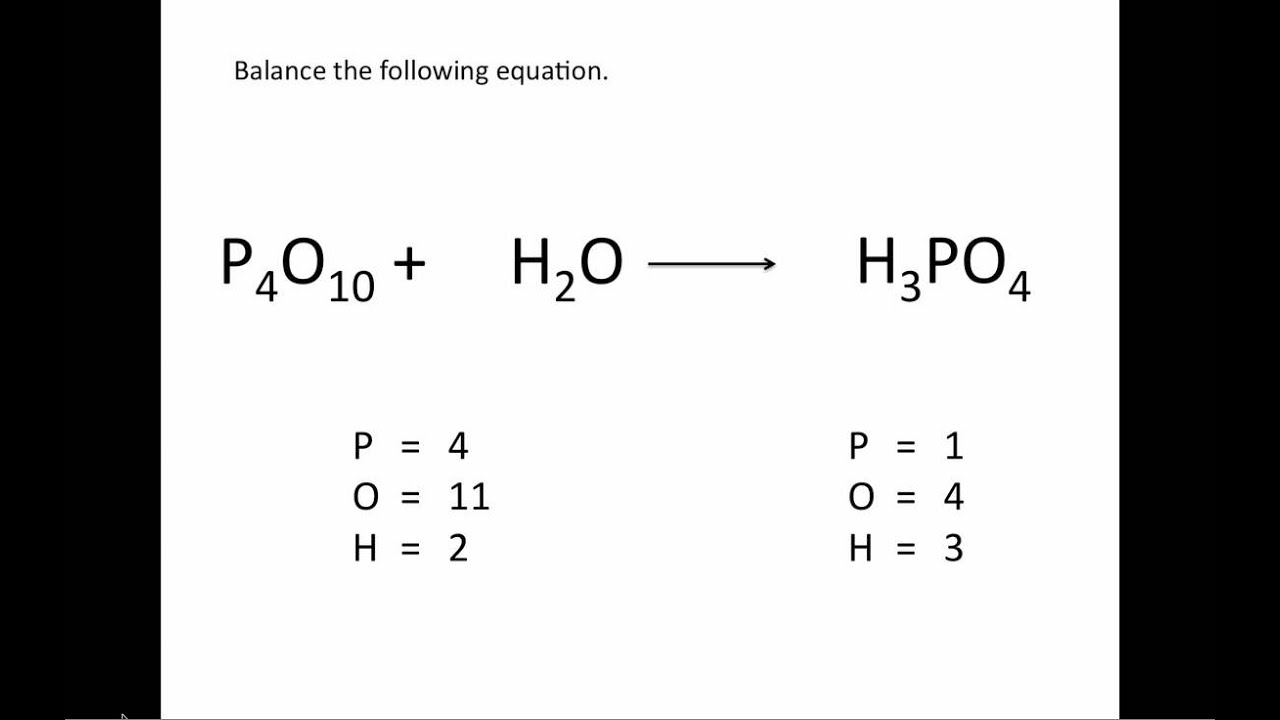
A balanced equation obeys the Law of Conservation of Mass, which states that matter is neither created nor destroyed in a chemical reaction. However, this equation isn't balanced because the number of atoms for each element is not the same on both sides of the equation. For example, in the reaction of hydrogen (H₂) with oxygen (O₂) to form water (H₂O), the chemical equation is: It shows the reactants (substances that start a reaction) and products (substances formed by the reaction).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
